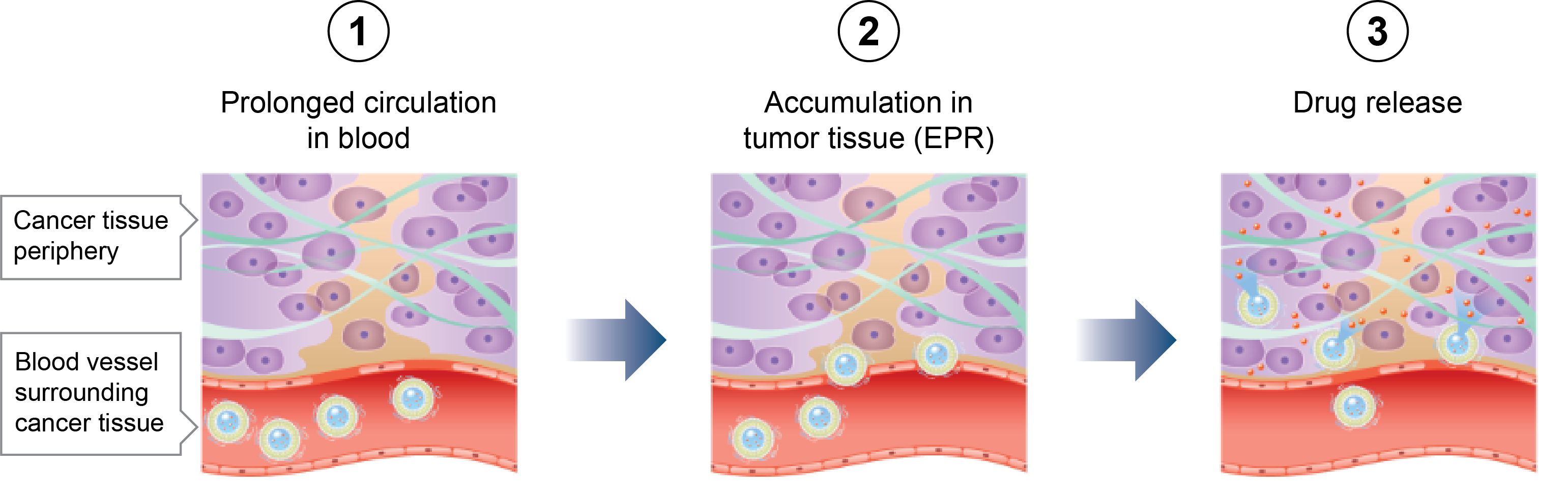
Peer-Reviewed Publications and Conference Presentation
| Pipeline | Category | Title | Link |
|---|---|---|---|
| FF-10850 | Conference | ASCO 2022: A first-in-human phase 1 dose escalation study of FF-10850 (liposomal topotecan) in patients with advanced solid tumors. | Journal of Clinical Oncology (ascopubs.org) | https://ascopubs.org/doi/abs/10.1200/JCO.2022.40.16_suppl.3101 |
| FF-10832 | Conference | ASCO 2022: A phase 1, first-in-human, dose-escalation and biomarker trial of liposomal gemcitabine (FF-10832) in patients with advanced solid tumors. | Journal of Clinical Oncology (ascopubs.org) | https://ascopubs.org/doi/abs/10.1200/JCO.2022.40.16_suppl.3097 |
| FF-10832 | Article | A physiologically based pharmacokinetic and pharmacodynamic model for disposition of FF-10832 – ScienceDirect | https://www.sciencedirect.com/science/article/pii/S0378517322008055 |
| FF-10832 | Article | A Liposomal Gemcitabine, FF-10832, Improves Plasma Stability, Tumor Targeting, and Antitumor Efficacy of Gemcitabine in Pancreatic Cancer Xenograft Models. Pharm Res. 2021 May 7. | |
| T-705 | Article | Vascular Leak and Hypercytokinemia Associated with Severe Fever with Thrombocytopenia Syndrome Virus Infection in Mice. Pathogens. 2019 Sep 21;8(4). pii: E158. | |
| T-705 | Article | Reevaluation of the efficacy of favipiravir against rabies virus using in vivo imaging analysis.Antiviral Res. 2019 Dec; 172:104641. | |
| T-705 | Article | Therapeutic effects of favipiravir against severe fever with thrombocytopenia syndrome virus infection in a lethal mouse model: Dose-efficacy studies upon oral administration. PLoS One. 2018 Oct 26;13(10):e0206416. | |
| T-705 | Article | Favipiravir (T-705) but not ribavirin is effective against two distinct strains of Crimean-Congo hemorrhagic fever virus in mice. Antiviral Res. 2018 Sep;157:18-26. | |
| T-705 | Article | Use of Favipiravir to Treat Lassa Virus Infection in Macaques. Emerg Infect Dis. 2018 Sep;24(9):1696-1699. | |
| T-705 | Article | Favipiravir (T-705) protects against Nipah virus infection in the hamster model. Sci Rep. 2018 May 15;8(1):7604. | |
| T-705 | Article | Effective Treatment of Experimental Lymphocytic Choriomeningitis Virus Infection: Consideration of Favipiravir for Use With Infected Organ Transplant Recipients. J Infect Dis. 2018 Jul 13;218(4):522-527. | |
| T-705 | Article | Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase. Proc Jpn Acad Ser B Phys Biol Sci. 93, 449-463. | |
| T-705 | Article | T-705 (Favipiravir) suppresses tumor necrosis factor α production in response to influenza virus infection: A beneficial feature of T-705 as an anti-influenza drug. Acta Virol. | |
| T-705 | Article | Modeling Severe Fever with Thrombocytopenia Syndrome Virus Infection in Golden Syrian Hamsters: Importance of STAT2 in Preventing Disease and Effective Treatment with Favipiravir. J Virol. 2017 Jan 18;91(3):e01942-16. | |
| T-705 | Article | Antiviral susceptibility of influenza viruses isolated from patients pre- and post-administration of favipiravi. Antiviral Res. 2016 Vol. 132 P170-177 | |
| T-705 | Article | The broad-spectrum antiviral favipiravir protects guinea pigs from lethal Lassa virus infection post-disease onset. Sci Rep. 2015 Oct 12;5:14775 | |
| T-705 | Article | In vitro activity of favipiravir and neuraminidase inhibitor combinations against oseltamivir-sensitive and oseltamivir-resistant pandemic influenza A (H1N1) virus. Arch Virol. 2014 Jun;159(6):1279-91. | |
| T-705 | Article | Favipiravir (T-705) protects against peracute Rift Valley fever virus infection and reduces delayed-onset neurologic disease observed with ribavirin treatment. Antiviral Res. 2014 Apr;104:84-92. | |
| T-705 | Article | Favipiravir (T-705) Inhibits Junín Virus Infection and Reduces Mortality in a Guinea Pig Model of Argentine Hemorrhagic Fever. PLOS Neglected Tropical Diseases 2013;7(12) e2614. | |
| T-705 | Article | Favipiravir (T-705), a novel viral RNA polymerase inhibitor. Antiviral Research 2013;100(2) 446–454. | |
| T-705 | Article | Synergistic combinations of favipiravir and oseltamivir against wild-type pandemic and oseltamivir-resistant influenza A virus infections in mice. Future Virology 2013;8(11):1085-1094. | |
| T-705 | Article | Mechanism of action of T-705 ribosyl triphosphate against influenza virus RNA polymerase. Antimicrob Agents Chemother. 2013;57(11):5202-8. | |
| T-705 | Article | Antiviral efficacy of favipiravir against two prominent etiological agents of hantavirus pulmonary syndrome. Antimicrob Agents Chemother. 2013;57(10):4673-80. | |
| T-705 | Article | Combinations of favipiravir and peramivir for the treatment of pandemic influenza A/California/04/2009 (H1N1) virus infections in mice. Antiviral Res. 2012;94(1):103-10. | |
| T-705 | Article | Effective oral favipiravir (T-705) therapy initiated after the onset of clinical disease in a model of arenavirus hemorrhagic Fever. PLoS Negl Trop Dis. 2011;5(10):e1342. | |
| T-705 | Article | Maporal virus as a surrogate for pathogenic New World hantaviruses and its inhibition by favipiravir. Antivir Chem Chemother. 2011;21(5):193-200. | |
| T-705 | Article | T-705 (Favipiravir) Inhibition of Arenavirus Replication in Cell Culture. Antimicrob Agents Chemother. 2011;55(2):782–7. | |
| T-705 | Article | In Vitro Antiviral Activity of Favipiravir (T-705) against Drug-Resistant Influenza and 2009 A(H1N1) Viruses. Antimicrob Agents Chemother. 2010;54(6):2517-24. | |
| T-705 | Article | Efficacy of favipiravir (T-705) and T-1106 pyrazine derivatives in phlebovirus disease models. Antiviral Res. 2010;86(2):121-7. | |
| T-705 | Article | T-705 (favipiravir) activity against lethal H5N1 influenza A viruses. Proc Natl Acad Sci USA 2010;107(2):882-7. | |
| T-705 | Article | Effects of the combination of favipiravir (T-705) and oseltamivir on influenza a virus infections in mice. Antimicrob Agents Chemother. 2010;54(1):126-33. | |
| T-705 | Article | Intracellular metabolism of favipiravir (T-705) in uninfected and influenza A (H5N1) virus-infected cells. J Antimicrob Chemother. 2009;64(4):741-6. | |
| T-705 | Article | In vitro and in vivo characterization of new swine-origin H1N1 influenza viruses. Nature 2009;460 (7258):1021-5. | |
| T-705 | Article | Effect of T-705 treatment on western equine encephalitis in a mouse model. Antiviral Res. 2009;82(3):169-71. | |
| T-705 | Article | T-705 (favipiravir) and related compounds: Novel broad-spectrum inhibitors of RNA viral infections. Antiviral Res. 2009;82(3):95-102. | |
| T-705 | Article | Activity of T-705 in a hamster model of yellow fever virus infection in comparison with that of a chemically related compound, T-1106. Antimicrob Agents Chemother. 2009;53(1):202-9. | |
| T-705 | Article | Efficacy of orally administered T-705 pyrazine analog on lethal West Nile virus infection in rodents. Antiviral Res. 2008;80(3):377-9. | |
| T-705 | Article | Treatment of late stage disease in a model of arenaviral hemorrhagic fever: T-705 efficacy and reduced toxicity suggests an alternative to ribavirin. PLoS One. 2008;3(11):e3725. | |
| T-705 | Article | In vitro and in vivo activities of T-705 against arenavirus and bunyavirus infections. Antimicrob Agents Chemother. 2007;51(9):3168-76. | |
| T-705 | Article | Efficacy of orally administered T-705 on lethal avian influenza A (H5N1) virus infections in mice. Antimicrob Agents Chemother. 2007;51(3):845-51. | |
| T-705 | Article | Mechanism of action of T-705 against influenza virus. Antimicrob Agents Chemother. 2005;49(3):981-6. | |
| T-705 | Article | In vitro and in vivo activities of T-705 and oseltamivir against influenza virus. Antivir Chem Chemother. 2003;14(5):235-41. | |
| T-705 | Article | In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob Agents Chemother. 2002;46(4):977-81. | |
| FF-10502 | Conference | ASCO 2019: Phase 1/2 trial of FF-10502-01, a pyrimidine antimetabolite, in patients with advanced cholangiocarcinoma and solid tumors. | https://ascopubs.org/doi/abs/10.1200/JCO.2019.37.15_suppl.3008 |
| FF-10502 | Conference | AACR 2018: A novel antimetabolite, FF-10502-01 exhibits potent antitumor activity via inhibition of both DNA replication and DNA damage repair in solid tumor cells | https://cancerres.aacrjournals.org/content/78/13_Supplement/326 |
| FF-10502 | Conference | AACR 2017: First-in-human phase 1 trial of pyrimidine anti-metabolite FF-10502-01 in patients with advanced cancer | https://cancerres.aacrjournals.org/content/77/13_Supplement/CT100 |
| FF-10502 | Article | FF-10502, an Antimetabolite with Novel Activity on Dormant Cells, Is Superior to Gemcitabine for Targeting Pancreatic Cancer Cells. J Pharmacol Exp Ther. 2018 Jul;366(1):125-135. | |
| FF-10850 | Conference | AAPS PharmaSci360 2019: Development of Pharmacokinetic-Pharmacodynamic Models for Tumor Growth Inhibition and Neutropenia after Administration of Liposomal Topotecan Injection, FF-10850 | |
| FF-10850 | Conference | AACR2019: FF-10850, a novel liposomal topotecan, achieved superior anti-tumor effects and a favorable safety profile via preferential distribution followed by dual payload release mechanisms in tumors | https://cancerres.aacrjournals.org/content/79/13_Supplement/1228 |
| FF-10832 | Conference | ASCO 2019: A phase I dose-escalation and immune biomarker study of intravenous FF-10832, liposomal gemcitabine, in patients with advanced solid tumors | https://ascopubs.org/doi/abs/10.1200/JCO.2019.37.15_suppl.TPS3163 |
| FF-10832 | Conference | AACR 2019: FF-10832 combined with immune checkpoint blockade shows favorable anti-tumor activity | https://cancerres.aacrjournals.org/content/79/13_Supplement/3953 |
| FF-10832 | Conference | AACR 2017: Liposomal gemcitabine, FF-10832, improves gemcitabine (GEM) pharmacokinetics (PK) and increases anti-tumor efficacy | https://cancerres.aacrjournals.org/content/77/13_Supplement/5148 |
| FF-10832 | Article | FF-10832 enables long survival via effective gemcitabine accumulation in a lethal murine peritoneal dissemination model. Cancer Sci. 2019 Sep;110(9):2933-2940. |